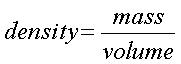Percent composition is the percentage by mass of a "species" in a chemical formula.
Before I tell you how to calculate percent composition, check out this video! It should help out a bit.
Percent Composition
To calculate the percent composition of a compound
- Calculate the molecular mass (molecular weight, formula mass, formula weight), MM, of the compound
- Calculate the total mass of each element present in the formula of the compound
- Calculate the percent compositon (percentage composition): % by weight (mass) of element
= (total mass of element present ÷ molecular mass) x 100
After you calculate the percentage of each compound, all of the percentages should add up to 100.
In this picture, it shows that there is 13% gravel and cobble, 30% salt and clay, and 57% sand in a sediment. This is an example of percent composition. Someone has already found out how much sand, salt, clay, gravel and cobble is in the sediment, and as you can see, they all add up to 100%.
Examples
1. Calculate the composition of C2H5OH.
Step 1:
- Calculate the Molar Mass of this object.
- Carbon has a mass of 12.0g. There are 2 atoms of Carbon in this substance, therefore the mass of carbon in this substance is 24.0g/mol.
- Hydrogen has a mass of 1.0g. There are 6 atoms of Hydrogen in this substance, therefore the mass of hydrogen in this substance is 6.0g/mol.
- Oxygen has a mass of 16.0g. There is one atom of Oxygen in this substance, therefore the mass of oxygen in this substance is 16.0g/mol.
- Add it all together to calculate the Molar Mass: 46g/mol.
Step 2:
- % of Carbon: 24.0g / 46.0g. This is the mass of carbon divided by the total Molar Mass. Then multiply it by 100. = 52.2%.
- % of Hydrogen: 6.0g / 46.0g. Multiplied by 100 = 13%.
- % of Oxygen: 16.0g / 46.0g. Multiplied by 100 = 34.8%.
2. If a compound contains 8.5g of F, 19.0g of Mg, and some amount of Be, and has a total mass of 41, calculate the % composition.
This question is harder because we have to figure out what the amount of Be is.
- The total MM is 41g. So add 8.5g and 19.0g to get 27.5. 41-27.5 = 13.5g of Be.
- MM of Fluoride: 8.5g
- MM of Magnesium: 19.0g
- MM of Beryllium: 13.5g
- % of F: 8.5g / 41g x 100 = 20.7%
- % of Mg: 19.0g / 41g x 100 = 46.3%
- % of Be: 13.5g / 41g x 100 = 33%
Pretty simple, right?
Just in case it still doesn't make sense, here is a step-by-step tutorial of an "Easy" example of Percent Composition.
Easy Example
If you're just breezing through this, here is a step-by-step tutorial of a "Harder" example of Percent Composition - made by the same guy!
Hard Example