Okay so cool, we finished our quiz on Molecular and Empirical Forumla, along with Percent Composition!
there isnt alot to say about this because we didnt have an actual lesson before or after the quiz...
I found that the quiz was quite straight forward! there was nothing on that quiz that Ms. Chen didnt
teach us. (im not being a suck up here)
Hopefully its not one of those quizzes where i thought i did well, but when i get it back... i didnt do so great.
If your still not getting it here are some VIDEOS:
Molecular and Empirical Formula: http://www.youtube.com/watch?v=gfBcM3uvWfs
and of course... A SONG, pretty intense: http://www.youtube.com/watch?v=slO9kN1XCnE
.... ok so they arent much good at singing, but if u look at the lyrics its about moles and molecular formula!
Percent Composition: http://www.youtube.com/watch?v=xbEeyT8nK84
ENJOY!
Wednesday, December 8, 2010
Monday, December 6, 2010
Review for Quiz!
Next class is quiz day!! so today we did a lot of review!
Quiz topics
ex. CO2
Total MM = 44 %compositions = mass of elements x 100%
MM of C = 12 mass of comp
MM of O = 32
% of C = 12g/mol x 100% = 27.3%
44g/mol
% of O = 32g/mol x 100% = 72.7%
44g/mol
talk through of CO2 composition
Empirical Formula
- lowest term ratio of atoms in formula
* All ionic compounds are empirical formula
We have 10.87 g of Fe and 4.66g of O, whats the empirical formula
#1 convert to moles
Fe 10.87g x 1mol = 0.195 moles
55.8g
O 4.66g x 1 mol = 0.291 moles
16g
#2 divide both by smallest molar amount
Fe = 1
O = 1.5
#3 scale to whole numbers
Fe = 2
O = 3
Molecular Formula
empirical formula of C2H5 molar mass of 58g/mol
MM C2H5 = 29g/molE
n = 58g/mol = 2
29 g
MF= 2( C2H5) = C4H10
really helpful video
New Stuff!!!
Empirical Formula of an organic compound
Found by
15.0g = 1 mol = 0.341
44g
8.18g = 1 mol = 0.454
18g
Molecules of C = 0.341 mol CO2 x 1mol C = 0.341 molecules 0.314 = 1 x 3 =3
1mol CO2 0.314
Molecules of H = 0.454 mol H2O x 2mol H = 0.908 molecules 0.908 = 2.66 x 3 = 8
1mol H2O 0.314
C3H8
Checking Mass
0.314 x 12g = 3.77
1mol 3.77 + 0.908 = 4.68g
0.908 x 1g = 0.908
1 mol
Study, Study, Study, Quiz next day!!
Quiz topics
- percent compositions
- empirical formula
- molecular formula
- calculating empirical formula of organic compounds
ex. CO2
Total MM = 44 %compositions = mass of elements x 100%
MM of C = 12 mass of comp
MM of O = 32
% of C = 12g/mol x 100% = 27.3%
44g/mol
% of O = 32g/mol x 100% = 72.7%
44g/mol
talk through of CO2 composition
Empirical Formula
- lowest term ratio of atoms in formula
* All ionic compounds are empirical formula
We have 10.87 g of Fe and 4.66g of O, whats the empirical formula
#1 convert to moles
Fe 10.87g x 1mol = 0.195 moles
55.8g
O 4.66g x 1 mol = 0.291 moles
16g
#2 divide both by smallest molar amount
Fe = 1
O = 1.5
#3 scale to whole numbers
Fe = 2
O = 3
Molecular Formula
empirical formula of C2H5 molar mass of 58g/mol
MM C2H5 = 29g/molE
n = 58g/mol = 2
29 g
MF= 2( C2H5) = C4H10
really helpful video
New Stuff!!!
Empirical Formula of an organic compound
Found by
- burning wood (reacting with CO20
- collecting and weighing the products
- from mass of a product the moles of each element in the original organic compound can be calculated
15.0g = 1 mol = 0.341
44g
8.18g = 1 mol = 0.454
18g
Molecules of C = 0.341 mol CO2 x 1mol C = 0.341 molecules 0.314 = 1 x 3 =3
1mol CO2 0.314
Molecules of H = 0.454 mol H2O x 2mol H = 0.908 molecules 0.908 = 2.66 x 3 = 8
1mol H2O 0.314
C3H8
Checking Mass
0.314 x 12g = 3.77
1mol 3.77 + 0.908 = 4.68g
0.908 x 1g = 0.908
1 mol
Study, Study, Study, Quiz next day!!
Thursday, December 2, 2010
Empirical + Molecular Formula
Today's lesson is on empirical and molecular formula.
Empirical Formula = gives the lowest term ratio of atoms (or moles of course) in the formula.
Ionic compounds are empirical formulas as well!
And here we have our example:
Lets say we have 10.87 g of Fe and 4.66 g of O. What is the empirical formula?
Well, first we convert grams to moles.
we want to convert the 10.87 grams so we will put that first. We want to eliminate grams so be sure to put the molar mass of the element in grams on the bottom.
10.87 g x 1 mole
------
55.8 g = 0.195 moles
4.66 g x 1 mole
-----
16 g = 0.291 moles
Now the smaller one of the two is the first one, so be use that one and divide by that amount.
0.195/0.195 = 1
0.291/0.195 = 1.5
Now we have to scale ratios to whole numbers. We need to get rid of that 1.5. We do that by multiplying both by two, because then the 1.5 turns into a 3!
Fe 1 x2 --> 2
O 1.5 x2 ---> 3
So the answer would be Fe2O3
Here is a problem for you to try for practice:
A compound used for sweetening is 57.14% C, 6.16% H, 9.52% N, and 27.18% O. Calculate the empirical formula of the sweetener and find the molecular formula. (The molar mass of the compound is 294.30 g/mol)
Remember to follow the steps!
1) convert to moles
2) divide both by the smallest molar amount
3) scale ratios
Now onto molecular formula:
Lets use this formula to help answer the questions:
n = molar mass of compound
--------------------------
molar mass of the empirical formula
The empirical formula of a gas is CH2. What is the molecular formula if the molar mass is 42 g/mol?
First, lets calculate the molar mass of the compound, because we need it in the equation.
The mass of carbon is 12, and the mass of hydrogen is 1, but there is 2 o them. So the molar mass of the compound is 14.
Now lets plug that into the equation!
n = 42 g/mol
--------
14 g/mol
which equals 3 g/mol.
Now write the answer 3(CH2)
make sure to distribute the 3.
so it is actually C3H6 = MF propane
Make sure you write the MF! It stands for molecular formula. You can remember it by thinking that it is Melanie's initals!
If you want a little extra help, I've got videos galore for you!
Molecular and Empirical Formula
A helpful chemistry tutorial
Extra help!
A super cool 16 minute tutorial! woah!
Empirical Formula = gives the lowest term ratio of atoms (or moles of course) in the formula.
Ionic compounds are empirical formulas as well!
And here we have our example:
Lets say we have 10.87 g of Fe and 4.66 g of O. What is the empirical formula?
Well, first we convert grams to moles.
we want to convert the 10.87 grams so we will put that first. We want to eliminate grams so be sure to put the molar mass of the element in grams on the bottom.
10.87 g x 1 mole
------
55.8 g = 0.195 moles
4.66 g x 1 mole
-----
16 g = 0.291 moles
Now the smaller one of the two is the first one, so be use that one and divide by that amount.
0.195/0.195 = 1
0.291/0.195 = 1.5
Now we have to scale ratios to whole numbers. We need to get rid of that 1.5. We do that by multiplying both by two, because then the 1.5 turns into a 3!
Fe 1 x2 --> 2
O 1.5 x2 ---> 3
So the answer would be Fe2O3
Here is a problem for you to try for practice:
A compound used for sweetening is 57.14% C, 6.16% H, 9.52% N, and 27.18% O. Calculate the empirical formula of the sweetener and find the molecular formula. (The molar mass of the compound is 294.30 g/mol)
Remember to follow the steps!
1) convert to moles
2) divide both by the smallest molar amount
3) scale ratios
Now onto molecular formula:
Lets use this formula to help answer the questions:
n = molar mass of compound
--------------------------
molar mass of the empirical formula
The empirical formula of a gas is CH2. What is the molecular formula if the molar mass is 42 g/mol?
First, lets calculate the molar mass of the compound, because we need it in the equation.
The mass of carbon is 12, and the mass of hydrogen is 1, but there is 2 o them. So the molar mass of the compound is 14.
Now lets plug that into the equation!
n = 42 g/mol
--------
14 g/mol
which equals 3 g/mol.
Now write the answer 3(CH2)
make sure to distribute the 3.
so it is actually C3H6 = MF propane
Make sure you write the MF! It stands for molecular formula. You can remember it by thinking that it is Melanie's initals!
If you want a little extra help, I've got videos galore for you!
Molecular and Empirical Formula
A helpful chemistry tutorial
Extra help!
A super cool 16 minute tutorial! woah!
Tuesday, November 30, 2010
Percent Composition
Today we learned about Percent Composition.
Percent composition is the percentage by mass of a "species" in a chemical formula.
Before I tell you how to calculate percent composition, check out this video! It should help out a bit.
Percent Composition
To calculate the percent composition of a compound
After you calculate the percentage of each compound, all of the percentages should add up to 100.
In this picture, it shows that there is 13% gravel and cobble, 30% salt and clay, and 57% sand in a sediment. This is an example of percent composition. Someone has already found out how much sand, salt, clay, gravel and cobble is in the sediment, and as you can see, they all add up to 100%.
Examples
1. Calculate the composition of C2H5OH.
2. If a compound contains 8.5g of F, 19.0g of Mg, and some amount of Be, and has a total mass of 41, calculate the % composition.
This question is harder because we have to figure out what the amount of Be is.
Pretty simple, right?
Just in case it still doesn't make sense, here is a step-by-step tutorial of an "Easy" example of Percent Composition.
Easy Example
If you're just breezing through this, here is a step-by-step tutorial of a "Harder" example of Percent Composition - made by the same guy!
Hard Example
Percent composition is the percentage by mass of a "species" in a chemical formula.
Before I tell you how to calculate percent composition, check out this video! It should help out a bit.
Percent Composition
To calculate the percent composition of a compound
- Calculate the molecular mass (molecular weight, formula mass, formula weight), MM, of the compound
- Calculate the total mass of each element present in the formula of the compound
- Calculate the percent compositon (percentage composition): % by weight (mass) of element
= (total mass of element present ÷ molecular mass) x 100
After you calculate the percentage of each compound, all of the percentages should add up to 100.
In this picture, it shows that there is 13% gravel and cobble, 30% salt and clay, and 57% sand in a sediment. This is an example of percent composition. Someone has already found out how much sand, salt, clay, gravel and cobble is in the sediment, and as you can see, they all add up to 100%.
Examples
1. Calculate the composition of C2H5OH.
Step 1:
- Calculate the Molar Mass of this object.
- Carbon has a mass of 12.0g. There are 2 atoms of Carbon in this substance, therefore the mass of carbon in this substance is 24.0g/mol.
- Hydrogen has a mass of 1.0g. There are 6 atoms of Hydrogen in this substance, therefore the mass of hydrogen in this substance is 6.0g/mol.
- Oxygen has a mass of 16.0g. There is one atom of Oxygen in this substance, therefore the mass of oxygen in this substance is 16.0g/mol.
- Add it all together to calculate the Molar Mass: 46g/mol.
Step 2:
- % of Carbon: 24.0g / 46.0g. This is the mass of carbon divided by the total Molar Mass. Then multiply it by 100. = 52.2%.
- % of Hydrogen: 6.0g / 46.0g. Multiplied by 100 = 13%.
- % of Oxygen: 16.0g / 46.0g. Multiplied by 100 = 34.8%.
2. If a compound contains 8.5g of F, 19.0g of Mg, and some amount of Be, and has a total mass of 41, calculate the % composition.
This question is harder because we have to figure out what the amount of Be is.
- The total MM is 41g. So add 8.5g and 19.0g to get 27.5. 41-27.5 = 13.5g of Be.
- MM of Fluoride: 8.5g
- MM of Magnesium: 19.0g
- MM of Beryllium: 13.5g
- % of F: 8.5g / 41g x 100 = 20.7%
- % of Mg: 19.0g / 41g x 100 = 46.3%
- % of Be: 13.5g / 41g x 100 = 33%
Pretty simple, right?
Just in case it still doesn't make sense, here is a step-by-step tutorial of an "Easy" example of Percent Composition.
Easy Example
If you're just breezing through this, here is a step-by-step tutorial of a "Harder" example of Percent Composition - made by the same guy!
Hard Example
Wednesday, November 24, 2010
The Mole
Sorry for the extremely late post, apparently when I thought I pressed publish post I just pressed preview, and never actually published it... -_-
Anyways, here's the lesson.
Today we learnt about moles!
And no, we aren't talking about the moles that are cute furry little creatures that live in the ground.
And no...we aren't talking about the little bumps on your face (that sometimes sprout hair. Yum).
A mole is a unit, just like m, km, g, L, etc. Pretty simple, right?
Here's a great video on the mole being a unit. Skip to about 0:30 to get the actual song!
A Mole is a Unit
It's pretty catchy if I do say so myself...that's going on my Ipod.
Now who came up with the mole? A man named Avogadro came up with it, and he also came up with a hypothesis and a number.
This man right here is Amedeo Avogadro.
His hypothesis was that two equal volumes of gas, at the same temperature and pressure, contain the same number of molecules.
If they have the same number of particles, the mass ratio is due to the mass of the particles.
Anyways, here's the lesson.
Today we learnt about moles!
And no, we aren't talking about the moles that are cute furry little creatures that live in the ground.
And no...we aren't talking about the little bumps on your face (that sometimes sprout hair. Yum).
A mole is a unit, just like m, km, g, L, etc. Pretty simple, right?
Here's a great video on the mole being a unit. Skip to about 0:30 to get the actual song!
A Mole is a Unit
It's pretty catchy if I do say so myself...that's going on my Ipod.
Now who came up with the mole? A man named Avogadro came up with it, and he also came up with a hypothesis and a number.
This man right here is Amedeo Avogadro.
His hypothesis was that two equal volumes of gas, at the same temperature and pressure, contain the same number of molecules.
If they have the same number of particles, the mass ratio is due to the mass of the particles.
Avogadro's Number:
The number of particles in 1 mole of any amount of substance is...
Now you may be thinking. WHAT? How am I supposed to memorize such a big number!?
It's actually a lot easier then you may think!
The mole is extremely important to chemists because it allows them to count atoms.
Here's another video on Mole Day!
Both of these songs I've posted are actually really helpful! Hahah I really like this one..
Too bad we already missed mole day. :'(
Tuesday, November 23, 2010
Even more mole conversions!!!
Last class we worked on the simpler mole conversions
- only going from mole to particle and reverse, and gram to mole and reverse
But!!! today we did harder mole conversions
- we went from particles to mass and reversed
(that means we had to convert through moles) yikes!!
The very first thing you need to know about harder mole conversions is the MOLE MAP!!
it's very helpful!!!
MM= molar mass
With this tool you can go through any conversions using moles!!
Here is a video simplifying mole conversions
Mole conversions made easy :)
Whenever your doing Mole conversions NEVER forget AVOGADRO'S number
(6.022x 10 ^23)
This is what we learned today and next class we have a quiz!!
If you want to try some practice questions here is a link to ms. Chens website
extra worksheets on ms chens website
- only going from mole to particle and reverse, and gram to mole and reverse
But!!! today we did harder mole conversions
- we went from particles to mass and reversed
(that means we had to convert through moles) yikes!!
The very first thing you need to know about harder mole conversions is the MOLE MAP!!
it's very helpful!!!
MM= molar mass
 |
| in each box all the top amounts are divided by the bottom amounts |
Here is a video simplifying mole conversions
Mole conversions made easy :)
Whenever your doing Mole conversions NEVER forget AVOGADRO'S number
(6.022x 10 ^23)
This is what we learned today and next class we have a quiz!!
If you want to try some practice questions here is a link to ms. Chens website
extra worksheets on ms chens website
Saturday, November 20, 2010
Mole Conversions
So, we have now started our new unit on the mole! yay!
Before we start, make sure you are comfortable calculating molar mass.
The first type of conversion is particles --> moles
Remember: 6.022 x10^23 particles/mole
Say you want to convert 3.01 x 10 ^24 particles of carbon to moles.
As with unit conversionis, you need the same units in the beginning of the equation, and on the bottom, so they can cancel eachother.
3.01 x 10^24 particles of carbon x 1 mole
______
6.022 x 10^23 particles
If you do the math, that means that there are 5.00 moles of carbon! Make sure you round off to the correct number of significant digits. The least number of sig figs in the equation is 3, therefore the answer should also have 3 sig figs.
Now, converting moles to molecules.
Lets try converting 0.75 moles of CO2 to atoms of oxygen.
Follow the exact same rules as given in the example above. Always follow those rules, these are just different units we are using.
0.75 moles x 6.022 x10^23 molecules
___________________
1 molecule
Once you multiply, you will calculate that the answer is 4.5 x 10^23 molecules CO2
We can also do conversions between moles and grams.
Lets calculate the grams of 2.04 moles of carbon
Use molar mass! The molar mass of Carbon is 12.0 grams/mol
Now lets do the math!
2.04 moles x 12.0 g
_____
1 mole = 24.5 grams of carbon
For compounds, to get the molar mass, just add up all the masses of each atom of each element.
Last but not least, grams to moles!
How about we convert 3.45 grams of carbon back to moles
Remember everything we have done so far! Find the mass of Carbon and plug it in. Then just follow the steps and do the math:
3.45 g x 1 mole
_____
12 grams and all this math = 0.288 moles of carbon
There you have it! How to convert from anything to everything! Hope this helps.
Before we start, make sure you are comfortable calculating molar mass.
The first type of conversion is particles --> moles
Remember: 6.022 x10^23 particles/mole
Say you want to convert 3.01 x 10 ^24 particles of carbon to moles.
As with unit conversionis, you need the same units in the beginning of the equation, and on the bottom, so they can cancel eachother.
3.01 x 10^24 particles of carbon x 1 mole
______
6.022 x 10^23 particles
If you do the math, that means that there are 5.00 moles of carbon! Make sure you round off to the correct number of significant digits. The least number of sig figs in the equation is 3, therefore the answer should also have 3 sig figs.
Now, converting moles to molecules.
Lets try converting 0.75 moles of CO2 to atoms of oxygen.
Follow the exact same rules as given in the example above. Always follow those rules, these are just different units we are using.
0.75 moles x 6.022 x10^23 molecules
___________________
1 molecule
Once you multiply, you will calculate that the answer is 4.5 x 10^23 molecules CO2
We can also do conversions between moles and grams.
Lets calculate the grams of 2.04 moles of carbon
Use molar mass! The molar mass of Carbon is 12.0 grams/mol
Now lets do the math!
2.04 moles x 12.0 g
_____
1 mole = 24.5 grams of carbon
For compounds, to get the molar mass, just add up all the masses of each atom of each element.
Last but not least, grams to moles!
How about we convert 3.45 grams of carbon back to moles
Remember everything we have done so far! Find the mass of Carbon and plug it in. Then just follow the steps and do the math:
3.45 g x 1 mole
_____
12 grams and all this math = 0.288 moles of carbon
There you have it! How to convert from anything to everything! Hope this helps.
Wednesday, November 17, 2010
Quiz Day
This class we had a quiz/test on experimental error and scientific notation.
It was pretty long.
Hopefully everyone did okay on it!
...Well I don't really have much to say.
Here's a review video on accuracy and precision.
Accuracy and precision
Since we didn't learn anything this class, here's a cute video of some guinea pigs eating corn.
SO CUTE.
K I'm done with being a loser now..
It was pretty long.
Hopefully everyone did okay on it!
...Well I don't really have much to say.
Here's a review video on accuracy and precision.
Accuracy and precision
Since we didn't learn anything this class, here's a cute video of some guinea pigs eating corn.
SO CUTE.
K I'm done with being a loser now..
Saturday, November 6, 2010
Accuracy/Precision and Measurement/Uncertainty
Accuracy and Precision
The textbook definitions (which are always useful) for accuracy and precision are:
-Precision is how reproducible a measurement is compared to other similar measurements. So how hard or easy is it to reproduce that measurement?
-Accuracy is how close the measurement (or average measurement) comes to the accepted or real value.
A good example of this would be a game we played in elementary school. We would play frisbee-golf where you have to throw the frisbee into the hulahoop (like a golf hole) and play the entire playground exactly like it is a golf course. So if you think about the hulahoop being the accepted or real value, accuracy is how close you can come to it when throwing the frisbee.
Measurement and Uncertainty
Every measure you will take or see is only an estimate, which has some degree of uncertainty.
Absolute uncertainty:
The uncertainty expressed in the units of measurement. There are 2 methods of doing this. Make sure to try both!
Method 1: Make at least 3 measurements. Calculate the difference between the average and the lowest or highest reasonable measurement. Be sure to include a +- sign at the beginning of the answer (I don't have that symbol on my computer)
Method 2: When making a measurement, always measure to the best precision you can (remember, precision is reproducibility!) Therefore, you should estimate to a fraction 0.1 of the smallest segment on the instrument scale.
Now, time for exactions! Here is how to calculate relative uncertainty:
Relative Uncertainty = absolute uncertainty / estimated measurement
Relative Uncertainty can be expressed in percentages or using significant figures.
Here is a video for your viewing pleasure, the guy really seems to know his chemistry stuff!
Super Cool Video
The textbook definitions (which are always useful) for accuracy and precision are:
-Precision is how reproducible a measurement is compared to other similar measurements. So how hard or easy is it to reproduce that measurement?
-Accuracy is how close the measurement (or average measurement) comes to the accepted or real value.
A good example of this would be a game we played in elementary school. We would play frisbee-golf where you have to throw the frisbee into the hulahoop (like a golf hole) and play the entire playground exactly like it is a golf course. So if you think about the hulahoop being the accepted or real value, accuracy is how close you can come to it when throwing the frisbee.
Measurement and Uncertainty
Every measure you will take or see is only an estimate, which has some degree of uncertainty.
Absolute uncertainty:
The uncertainty expressed in the units of measurement. There are 2 methods of doing this. Make sure to try both!
Method 1: Make at least 3 measurements. Calculate the difference between the average and the lowest or highest reasonable measurement. Be sure to include a +- sign at the beginning of the answer (I don't have that symbol on my computer)
Method 2: When making a measurement, always measure to the best precision you can (remember, precision is reproducibility!) Therefore, you should estimate to a fraction 0.1 of the smallest segment on the instrument scale.
Now, time for exactions! Here is how to calculate relative uncertainty:
Relative Uncertainty = absolute uncertainty / estimated measurement
Relative Uncertainty can be expressed in percentages or using significant figures.
Here is a video for your viewing pleasure, the guy really seems to know his chemistry stuff!
Super Cool Video
Thursday, November 4, 2010
Lab Day!
Today we did a Lab on the thickness of aluminum foil!!
I know it doesn't sound very interesting but it was actually pretty interesting how to do it!
With this data we collected we had to use formulas for density, and volume
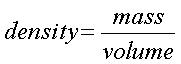
Volume = (length)(width)(height)
*height is our thickness that we were originally trying to find
Here is a video on how to find the thickness of aluminum foil using these formulas=
http://www.mefeedia.com/watch/29045208
Next class we have a quiz on this lab!!
I know it doesn't sound very interesting but it was actually pretty interesting how to do it!
- We took 3 almost square pieces of foil.
- We measured all for sides and came up with an average lengh and width of 1 piece of foil.
- We got to use a centigram balance to find the mass of each piece of foil
 | |||
| centigram balance ( ours were much older) |
With this data we collected we had to use formulas for density, and volume
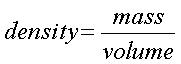
Volume = (length)(width)(height)
*height is our thickness that we were originally trying to find
Here is a video on how to find the thickness of aluminum foil using these formulas=
http://www.mefeedia.com/watch/29045208
Next class we have a quiz on this lab!!
Thursday, October 28, 2010
Significant Figures! (sig figs)
what are significant figures you say?
for instance, in the measurement 7.63cm "3" is uncertain and "7 and 6" are certain digits.
To count the number of significant figures you would have to include all the digits as well as the uncertain digit! To help find the number of sig figs here are a few pointers:
a)count all the numbers starting at the first non-zero digit on the left
b)for a number written in scientific notation count the digits in the coefficient
ex 1. 2.43 > 3 sig figs
2. 5.254 > 4 sig figs
Details:
Exact Numbers
Rounding
Adding and Subtracting
Multiplying and Dividing
well...
- they are often used in connection to rounding
- establish the value of a number
Significant Digits
In a measurement, the last digit is known to be an uncertain digit, because it may turn out to be one digit higher or lower. You will never know!for instance, in the measurement 7.63cm "3" is uncertain and "7 and 6" are certain digits.
To count the number of significant figures you would have to include all the digits as well as the uncertain digit! To help find the number of sig figs here are a few pointers:
a)count all the numbers starting at the first non-zero digit on the left
b)for a number written in scientific notation count the digits in the coefficient
ex 1. 2.43 > 3 sig figs
2. 5.254 > 4 sig figs
Details:
- Zeroes placed before other digits are not significant; 0.0095 has two sig figs
- Zeroes placed after other digits but behind a decimal point are significant; 7.090 has four sig figs
- Zeroes at the end of a number are significant only if they are behind a decimal point
- Zeros placed after other digits without a decimal point are not counted; 8500 has two sig figs
Exact Numbers
Not every number is measured experimentally, some numbers are exact. They have an infinite number of significant digits and no rounding is needed.
ex 1. a pair of earings; there cannot be 1.92 earings rounds to 2
2. 360 degrees in a circle
3. 1m = 100cm
Rounding
Rounding is replacing the numbers value that is approximately equal but has a shorter or simpler representation.
- Determine what your rounding digit is and look to the right side of it.
ex. When asked to round to the closest tens - your rounding digit is the second number to the left (ten's place) - If that digit is greater than 5, round one up
- If that digit is lower the 5, dont change it, keep it the same
- If that digit is equal to 5 and there are more numbers that are not zeros following it, round one up
- If that digit is equal to 5 but ends at 5, round the digit before that to an even number (0,2,4)
ex. round to the nearest 10th
a) 74.24 = 74.2
b) 7.273 = 7.3
c) 67.35 = 67.4
d) 2.652 = 2.7
Adding and Subtracting
When adding and subtracting, the number of decimal places in the ANSWER should be the SAME as the number with the LEAST decimal places in any of the terms being added or subtracted.
2.548g (3 decimal places)
5.72g (2 decimal places)
+1.1g (1 decimal place)
9.36g
the answer is: 9.4g because the lowest decimal place in the equation is 1; also DON'T FORGET TO ROUND IT!
Multiplying and Dividing
When multiplying or dividing numbers, the number of decimal points/significant figures in the RESULT is the same as the LEAST number of decimal points/significant figures in any of the multiplied or divided terms.
12.54mm (2 decimal points)
x 1.3mm (1 decimal point)
16.302mm2
the answer is: 16.3mm2 because the lowest decimal place in the equation is 1; and then ROUND IT!
I know its alot to take in! but practice makes perfect! just keep going over the rules and you'll get it!
ENJOY
Wednesday, October 20, 2010
Naming Acids!
Naming acids is really quite easy.
First off, what are acids?
Acids are formed when a compound composed of hydrogen ions and a negatively charged ion are dissolved in water. This is a state called aqueous. (aq.)
Ions separate when dissolved in water.
So now that we know what acids are, on to how to name them!
With simple acids, all you have to do is remember these 3 simple steps.
For example, HCN. You may think that this is Hydrogen Cyanide. But if you take the steps as shown above, you'll now know that it's really Hydrocyanic Acid.
Just in case this isn't really clicking for you, try checking out this video.
First off, what are acids?
Acids are formed when a compound composed of hydrogen ions and a negatively charged ion are dissolved in water. This is a state called aqueous. (aq.)
Ions separate when dissolved in water.
So now that we know what acids are, on to how to name them!
With simple acids, all you have to do is remember these 3 simple steps.
- Use "hydro" at the beginning.
- Drop the last syllable of the non-metal and replace it with "-ic".
- Add "acid" at the end.
Pretty easy, right?
Let's try an example.
HF, which you know as Hydrogen Fluoride, now becomes Hydrofluoric Acid.
See what I did there? I changed the end of Fluoride, by replacing the "-ide" with "-ic", and then added Acid at the end. It's as simple as that!
Let's try naming complex acids now.
Once again, there are only 3 rules you need to know for naming complex acids.
- Replace "-ate" with "-ic".
- Replace "-ite" with "-ous".
- Add "acid" at the end.
This may seem like something thats hard to remember, but it's really not.
Here's something that Mr. Morrison taught Ms. Chen to teach us!
"We ate ic-y sushi and got appendic ite-ous."
Get it? You replace ate with ic, and ite with ous.
For example, HCN. You may think that this is Hydrogen Cyanide. But if you take the steps as shown above, you'll now know that it's really Hydrocyanic Acid.
Now a harder one...
HNO2.
Pfft, harder? That's easy!
Hydronitrite Acid? Right?
Wrong. With more complex acids, you need to DROP THE HYDRO.
The answer for this one is actually Nitrous Acid. "-ous", because the original ion ends with "-ite". Then just add acid at the end!
The only two elements that do not lose the last part of their names are Sulphur and Phosphur.
They change to Sulphuric, Phosphurous, etc.
Just in case this isn't really clicking for you, try checking out this video.
Hope this makes sense for you!
Tuesday, October 19, 2010
Lab Day!
Last class we looked at the different separation techniques! This class we did an experiment on chromatography!!
Our Lab today was called Separation of a Mixture by Paper Chromatography
Key words
Preparation
Starting the experiment
Now it's time for the calculation part of the experiment!!!
Here is a video of another chromatography experiment!
It is different but the results are very similar to that of the one we did!
http://www.youtube.com/watch?v=Oy_yTOwyRLM&feature=related
Results
In this video you saw the separation of the ink, our results were the same. ours however separated into the primary colours. Our unknown separated into all 3 primary colours ( red, yellow, blue) and our green separated into yellow and blue.
This was our Lab of the day!
Next day we have our first test!!!
(ahhhhhhhh!!!)
Our Lab today was called Separation of a Mixture by Paper Chromatography
Key words
- solute front ( ie. food coloring)
- solvent front (ie. water)
- Rf values (d1 over d2)
- d1 (distance traveled by solute)
- d2 (distance traveled by solvent)
Preparation
- The first step in this lab, was of course to put all of our safety equipment on!
- obtain 3 large test, and 3 Erlenmeyer flasks.
- next we had to get 3 strips of paper, 22 cm each. 4 cm from the end we drew a line and trimmed the edge to make a point.
- we placed water 2 cm deep in the test tube.
Starting the experiment
- On our first piece of paper we placed either a red, yellow, or blue dot on the line we drewin the preparation stage.
- we placed the strip of paper in a test tube (The flat surfaces are not aloud to touch the test tube)
- Here we wait for 10 min writting notes on what happens
- because that process takes so long, we moved on to part III of the experiment
- take another of our strip and put a dot of green food colouring on the line
- take our last strip and put the unknown mixture on the line
- put both of the strips of paper in the last 2 test tubes
- After 20 mins and you are fully satisfied that nothing more will change, you remove all the strips from the test tubes
Now it's time for the calculation part of the experiment!!!
- when you've taken the strips out measure the distance traveled by the solute (label d1), and the solvent (label d2)
- with your results now it's time to find the Rf value
- to calculate this you use the formula ( d1 divided by d2= Rf)
Here is a video of another chromatography experiment!
It is different but the results are very similar to that of the one we did!
http://www.youtube.com/watch?v=Oy_yTOwyRLM&feature=related
Results
In this video you saw the separation of the ink, our results were the same. ours however separated into the primary colours. Our unknown separated into all 3 primary colours ( red, yellow, blue) and our green separated into yellow and blue.
This was our Lab of the day!
Next day we have our first test!!!
(ahhhhhhhh!!!)
Sunday, October 17, 2010
Different Techniques for Seperation
Today we learned about different techniques to seperate mixtures. Sounds fun, right? Well, it is!
FYI: The more similar the properties are, the more difficult it is to seperate them
The basic techniques are:
-Filtration
-Flotation
-Crystallization
-Chromotography
-Distillation
Hand seperation is useful for mechincal or heterogeneous mixtures. You can boil away the liquid and the solid will remain. (Like boiling away water in a pot of pasta)
Filtration is useful for solids that haven't been dissolved and liquids. It passes a mixture that contains solid particles through a porous filter. (this can sometimes we done for drinking water) You can also use filter paper - residue left in the filter paper and the filtrate goes through the filler paper
Crystallization solids are seperated by filtration or flotation and the solid comes out as pure crystals. Crystals are then filtered from the remaining solvent.

Gravity Seperation is for solids based on density. A centrifuge (device) whirls a test tube around at high speed forcing denser materials for to the bottom. But this works best for small volumes only.


FYI: The more similar the properties are, the more difficult it is to seperate them
The basic techniques are:
-Filtration
-Flotation
-Crystallization
-Chromotography
-Distillation
Hand seperation is useful for mechincal or heterogeneous mixtures. You can boil away the liquid and the solid will remain. (Like boiling away water in a pot of pasta)
Filtration is useful for solids that haven't been dissolved and liquids. It passes a mixture that contains solid particles through a porous filter. (this can sometimes we done for drinking water) You can also use filter paper - residue left in the filter paper and the filtrate goes through the filler paper
Crystallization solids are seperated by filtration or flotation and the solid comes out as pure crystals. Crystals are then filtered from the remaining solvent.

Crystallized Snow
Gravity Seperation is for solids based on density. A centrifuge (device) whirls a test tube around at high speed forcing denser materials for to the bottom. But this works best for small volumes only.

A centrifuge can be used at university
video on centrifuges (Super cool experiment)
Solvent extraction is when a component moves into a solvent shaken with the mixture. It works best with solvents that dissolve only one component. For mechanical mixtures (solid and solid - like nickels and dimes) you can use liquid to dissolve one solid but not the other. That way, the solid you want is left behind or has dissolved.
Distillation is used for liquid in a liquid solution. Most bottled water you find will have been distilled to seperate any sort of dirt or bacteria from the water. You can heat the mixture to cause the low-boiling components to vapourize, and that way the volatilized components have been collected and condensed.

Evian (being modeled by a mermaid?) distills their water
Chromotography can seperate more complex mixtures like drugs and plastic and the analyses can be highly accurate. It is when a mobile phase sweeps the sample over a stationary phase to seperate the two. An example would be when the wind sweeps your wig to reveal that you are bald! (Like in the trident commercial) There are two types of chromotography, sheet chromotography and thin layer chromotography.
Monday, October 11, 2010
Writing and Naming Ionic and Covalent Compounds
What are Ionic Compounds?
Well, basically they are compounds where two or more ions are held together by electric attraction.
One of the ions are positively charged, and the other is negatively charged. When writing the compounds, always remember that the positively charged ion (metal) is always first!
To make life easier, looking at the periodic table, the positively charged ions (metals) are on the left side and the negatively charged ions (non-metals) are on the right side.
Writing and Naming Ionic Compounds
2)Write the charges
3)Cross over the charges from top to bottom
4)Remove the chages
In example 1 we took their charges and "cross-over" from top to bottom. Remember, before writing the formula down make sure that charges are correct and the metal is written first. The reason we do this is because we want the resulting charge to equal zero!
Ex 2. You will find yourself coming across an element that has 2 charges instead of 1, but dont panic! turn that frown upside down, it only takes one more easy step :)
For instance, take copper (Cu), it has a charge of +2 and +1. Just follow the same steps as example 1, but make sure you use roman numerals to tell us which charge you are using.
a) Copper (I) oxide = Cu2O
b) SnCl4 = Tin (IV) Cloride
Also, under Ionic compounds are Complex ions: A group of atoms that behave as one atom.
You can find these ions behind the periodic table Ms. Chen distributed :)
*Writing these formulas only requires the same steps as the one given before.The underlined elements are complex ions and are found in the back of the periodic table or the picture shown above.
a) Calcium Hydroxide = Ca(OH)2 < when dealing with complex ions, put brackets to show the 2 goes into both O and H
b) Copper (II) Nitrite = Cu(NO2)2
What are Covalent Compounds?
It is a compound in which two atoms bond, they share electrons rather than transfer. Covalent compounds are formed when two non-metals bond to each other whereas ionic compounds form when metals bond to non-metals.
Diatomic Elements is another thing you got to watch out for in covalent compounds. They are a special group of molecules that consist of 2 identical atoms, which are:
H2, O2, F2, Br2, I2, N2, Cl2
In covalent compounds we use greek prefixes to indicate the number of atoms
*memorize this for tests in the furture*
Mono - 1 Penta - 5 Octa - 8
Di - 2 Hexa - 6 Nona - 9
Tri - 3 Hepta - 7 Deca - 10
Tera - 4
Writing and Naming Ionic Compounds:
*The cross over process is not needed for writing ionic formulas
Ex a) CO2 = Carbon dioxide
b) N2O4 = Dinitrogen tetroxide
c) Sulphur trioxide = SO3
VIDEOS! For all you visual learners, like me :)
Ionic Compounds: http://www.youtube.com/watch?v=vscoYh6m46M
Covalent Compounds: http://www.youtube.com/watch?v=fxE1KOeNZXw
Enjoy :)
Well, basically they are compounds where two or more ions are held together by electric attraction.
One of the ions are positively charged, and the other is negatively charged. When writing the compounds, always remember that the positively charged ion (metal) is always first!
To make life easier, looking at the periodic table, the positively charged ions (metals) are on the left side and the negatively charged ions (non-metals) are on the right side.
Writing and Naming Ionic Compounds
The Cross over rule:
1)Write the symbols2)Write the charges
3)Cross over the charges from top to bottom
4)Remove the chages
 | ||||
Ex 2. You will find yourself coming across an element that has 2 charges instead of 1, but dont panic! turn that frown upside down, it only takes one more easy step :)
For instance, take copper (Cu), it has a charge of +2 and +1. Just follow the same steps as example 1, but make sure you use roman numerals to tell us which charge you are using.
a) Copper (I) oxide = Cu2O
b) SnCl4 = Tin (IV) Cloride
Also, under Ionic compounds are Complex ions: A group of atoms that behave as one atom.
You can find these ions behind the periodic table Ms. Chen distributed :)
*Writing these formulas only requires the same steps as the one given before.The underlined elements are complex ions and are found in the back of the periodic table or the picture shown above.
a) Calcium Hydroxide = Ca(OH)2 < when dealing with complex ions, put brackets to show the 2 goes into both O and H
b) Copper (II) Nitrite = Cu(NO2)2
What are Covalent Compounds?
It is a compound in which two atoms bond, they share electrons rather than transfer. Covalent compounds are formed when two non-metals bond to each other whereas ionic compounds form when metals bond to non-metals.
Diatomic Elements is another thing you got to watch out for in covalent compounds. They are a special group of molecules that consist of 2 identical atoms, which are:
H2, O2, F2, Br2, I2, N2, Cl2
In covalent compounds we use greek prefixes to indicate the number of atoms
*memorize this for tests in the furture*
Mono - 1 Penta - 5 Octa - 8
Di - 2 Hexa - 6 Nona - 9
Tri - 3 Hepta - 7 Deca - 10
Tera - 4
Writing and Naming Ionic Compounds:
*The cross over process is not needed for writing ionic formulas
Ex a) CO2 = Carbon dioxide
b) N2O4 = Dinitrogen tetroxide
c) Sulphur trioxide = SO3
VIDEOS! For all you visual learners, like me :)
Ionic Compounds: http://www.youtube.com/watch?v=vscoYh6m46M
Covalent Compounds: http://www.youtube.com/watch?v=fxE1KOeNZXw
Enjoy :)
Tuesday, October 5, 2010
Lab Day!
Today was lab day!
Today's lab was The Heating and cooling curves of a pure substance
The pure substance we used today was - Dodecanoic Acid
ps. it did not smell good, it made me feel dizzy!
Like all labs our first step was to put on our lab apron and safety goggles
This is one of the most important steps, because you always have to put safety first!
Our objective for the lad was to:
- decide which role you'll play (one person observes and the other records the data)
- next was to obtain our materials and our acid
Materials
This was all the setting up part, next is the fun stuff!!
In our textbook it said to do the heating process first, but instead we did the cooling process first.
This picture represents what our set up looked like, although there was water in the beaker and a test tube in the clamp above the beaker.
It took us around 9 min for the dodecanoic acid to drop from 43'C to 25'C and 13 min for the temperature to get back up and reach 50'C.
We had to complete a table of the temperature at every 30 seconds and observations at every step, then we had to graph it.
It was a lot of Hard work, but it was a really cool experiment!
Today's lab was The Heating and cooling curves of a pure substance
The pure substance we used today was - Dodecanoic Acid
ps. it did not smell good, it made me feel dizzy!
Like all labs our first step was to put on our lab apron and safety goggles
This is one of the most important steps, because you always have to put safety first!
Our objective for the lad was to:
- investigate the heating process for solid dodecanoic acid
- investigate the cooling process for liquid dodecanoic acid
- determine and compare the melting and freezing points of dodecanoic acid
- decide which role you'll play (one person observes and the other records the data)
- next was to obtain our materials and our acid
Materials
- ring stand
- buret clamp
- hot plate
- test tube
- beaker
- thermometer
This was all the setting up part, next is the fun stuff!!
In our textbook it said to do the heating process first, but instead we did the cooling process first.
This picture represents what our set up looked like, although there was water in the beaker and a test tube in the clamp above the beaker.
It took us around 9 min for the dodecanoic acid to drop from 43'C to 25'C and 13 min for the temperature to get back up and reach 50'C.
We had to complete a table of the temperature at every 30 seconds and observations at every step, then we had to graph it.
It was a lot of Hard work, but it was a really cool experiment!
- The acid went completely solid and it turned white ( it started as a pale yellow clear color)
- Then during the heating process it turned to a white goo before it went to its original state.
Saturday, October 2, 2010
Textbook Summary
Summarizing for a part of the reading (25-34) from the textbook:
What are chemists?
Chemists specialize in matter:
A mixture is two or more kinds of matter that have separate identities. It is easily separated into component parts, and some people say it is impure.
Mixtures that CAN be separated:
Check out this video called the Atom Song!
Skip to 30 seconds to get to the song.
The Atom Song!
Summarizing for you a part of the reading (36-39) from the textbook:
The general idea here is that matter is made of atoms.
The idea that matter is made of atoms did not become popular until the early 1800s.
Now, lets look at some terms:
Atom: The smallest possible piece of something, they vary in size and mass
Elements: Pure substances that cannot be broken down
Solid: Holds its shape since atoms are stuck together
Liquid: The temperature is increased and atoms will vibrate and flow past one another
Molecule: particles made up of more than one atom
Compounds: Made by combining elements in definite proportions (made of 2 or more types of atoms)
Ions: particles that have an electrical charge
Ionic: melt to form ions
Molecular: melt as molecules
My Socials Teacher once told me that once you know the meaning of the terms, it makes the material much easier to learn. So hopefully this helps!
Here's a cool video about atoms and molecules, and the guy talking has braces! It's pretty funny.
Click here
What are chemists?
Chemists specialize in matter:
- what it is
- how one kind differs form another
- what different kinds have in common
- how one kind can be changed to another
- how it can be kept the same
Water is one of the most familiar kinds of matter. The temperature at which matter changes from a liquid to a gas is called boiling point.
A mixture is two or more kinds of matter that have separate identities. It is easily separated into component parts, and some people say it is impure.
Mixtures can be heterogeneous mixtures or homogeneous mixtures.
Heterogeneous mixtures- the substances in the kind of a mixture are not spread out evenly.
Example: Salad Dressing
Homogeneous mixtures- the substances are spread evenly throughout, a homogeneous mixture is called a solution.
Example: Vinegar
By adding things to pure water, you can quickly find that there are no mixtures that do not scatter light.
If table salt is added, it disappears. You can taste it, but it cannot be seen.
Therefore, the table salt does not scatter light.
Therefore, the table salt does not scatter light.
Solutions - mixtures that look uniform throughout and do not scatter light.
Distillation - separates most mixtures.
Mixtures that CAN be separated:
- household ammonia
- alcoholic beverages - for example, whiskey
Pure Substances have a constant boiling point. Mixtures ordinarily do not.
Freezing Point - the temperature at which a liquid changes to a solid
Melting Point - the temperature at which a solid changes to a liquid
Density - a property of matter that describes its mass per unit volume
Chemical Changes - changes that produce a new kind of matter with different properties
Physical Changes - changes that change the appearance, but do not produce new kinds of matter
Decomposition - when one kind of matter comes apart (decomposes) to form two or more kinds of matter
Electrolysis - involves passing an electric current through a substance, causing it to decompose into new kinds of matter
The Law of Definite Composition: A compound always contains two or more elements combined in a definite proportion by mass
The Law of Multiple Proportions: The fact that two or more compounds with different proportions of the same elements can be made.
Check out this video called the Atom Song!
Skip to 30 seconds to get to the song.
The Atom Song!
Summarizing for you a part of the reading (36-39) from the textbook:
The general idea here is that matter is made of atoms.
The idea that matter is made of atoms did not become popular until the early 1800s.
Now, lets look at some terms:
Atom: The smallest possible piece of something, they vary in size and mass
Elements: Pure substances that cannot be broken down
Solid: Holds its shape since atoms are stuck together
Liquid: The temperature is increased and atoms will vibrate and flow past one another
Molecule: particles made up of more than one atom
Compounds: Made by combining elements in definite proportions (made of 2 or more types of atoms)
Ions: particles that have an electrical charge
Ionic: melt to form ions
Molecular: melt as molecules
A very large water molecule
A caffeine molecule
LSD (acid) molecule. Don't try this at home kids!
My Socials Teacher once told me that once you know the meaning of the terms, it makes the material much easier to learn. So hopefully this helps!
Here's a cool video about atoms and molecules, and the guy talking has braces! It's pretty funny.
Click here
Thursday, September 30, 2010
Lab Day!
So last class we did a lab! about physical and chemical change.
To help us really understand how the lab was going to work Ms. Chen had us make our own flow chart,
consisting of all the steps we had to do in order to make the lab work properly.
Remember always tie any long hair up, wear close toed shoes, and wear lab glasses at all times during a lab until the process is finished! we wouldn't want anything bad happening!
The equipment used were:
As a result, when the solutions were combined they either changed in color or created tiny bubbles.
From this we were to figure out weather the mixed solutions had a Physical Change or a Chemical Change.
To decide which one it is here's something to help you out:
1. A physical change is reversible, a chemical change is not. For example, the freezing of water would be a physical change because it can be reversed, whereas the burning of wood is a chemical change - you can't 'unburn' it
2. A physical change is a change in which no new substance is formed; a chemical change results in the formation of one or more new substances.
Still dont get it? or are a visual learner, check out this vid!
http://www.youtube.com/watch?v=oQnPNBVmwuU&feature=related
Enjoy!
To help us really understand how the lab was going to work Ms. Chen had us make our own flow chart,
consisting of all the steps we had to do in order to make the lab work properly.
Remember always tie any long hair up, wear close toed shoes, and wear lab glasses at all times during a lab until the process is finished! we wouldn't want anything bad happening!
The equipment used were:
- 4 test tubes, labelled A,B,C and D
- a test tube rack
- 4 medicine droppers
- glass square
As a result, when the solutions were combined they either changed in color or created tiny bubbles.
From this we were to figure out weather the mixed solutions had a Physical Change or a Chemical Change.
To decide which one it is here's something to help you out:
1. A physical change is reversible, a chemical change is not. For example, the freezing of water would be a physical change because it can be reversed, whereas the burning of wood is a chemical change - you can't 'unburn' it
2. A physical change is a change in which no new substance is formed; a chemical change results in the formation of one or more new substances.
Still dont get it? or are a visual learner, check out this vid!
http://www.youtube.com/watch?v=oQnPNBVmwuU&feature=related
Enjoy!
Monday, September 27, 2010
Quiz Day
Today (unfortunately) was quiz day!
but before we wrote our quizzes on Conversion we had a review on Matter!
What is Matter??
- it is anything with mass and volume
ex. pretty much everything you've ever known about (you, your clothes, your house, air, trees, animals etc.)
- Matter is made up of 2 parts: Pure substances and mixtures
This is were things get a little trickier!
Pure substances have only 1 set of properties however mixtures have more then 1
Within Pure substances there are elements and compounds
-we learned a lot about elements and compounds throughout grades 6 to 10
-just to sum it up, elements are in there simplest forms, and compounds are made of elements
Mixtures were a pretty new idea today, to be honest I cant really remember learning about them in the past.
homogeneous and heterogeneous are the 2 ways mixtures are physically combined.
that probably makes no sense right now (I thought so at first) but really it's not that hard
Homogeneous basically means you cant tell how many components there are, you can only see 1
- ex. milk or fog
 | |
| Milk: only 1 visible component |
Heterogeneous is the opposite, it is obvious that it has more then 1 component
-ex. water + oil mixed together
 | |
| water + oil: more then 1 visible component |
This seems like a lot of information, but I'm still not done!
Another thing we learned before our quiz was the difference between Physical and Chemical change, instead of telling you about it, here is a video that tells you about the 2 changes
I hope you enjoyed the video, with it's little song!!
Thursday, September 23, 2010
Thursday, September 23rd, 2010
It's the long weekend this weekend! So today, the Happy Halogens worked on getting ready for our quiz on unit conversions for Monday. Here are some helpful resources so that you can feel confident and prepared to get that A+!
Using Julia's instructions, here are some extra practice questions to work on:
Questions
Answers
Also, if you want to try something just for fun, use this to convert anything you want to anything you want!
If you are still lost and confused and don't understand what is going on or how to do it (I know the feeling) this video will help clear up any problems:
The Greatest Unit Conversion Video Ever Made!
Enjoy!
Using Julia's instructions, here are some extra practice questions to work on:
Questions
Answers
Also, if you want to try something just for fun, use this to convert anything you want to anything you want!
If you are still lost and confused and don't understand what is going on or how to do it (I know the feeling) this video will help clear up any problems:
The Greatest Unit Conversion Video Ever Made!
Enjoy!
Tuesday, September 21, 2010
Scientific Notation.
It may look hard, but once you get the hang of it...it isn't hard at all.
Basically, it's used to express very large or very small numbers, using powers of 10.
It's just a shorter method.
Instead of writing:
It may look hard, but once you get the hang of it...it isn't hard at all.
Basically, it's used to express very large or very small numbers, using powers of 10.
It's just a shorter method.
Instead of writing:
we write:
The first number, 7.53, is called the coefficient.
It must be in between 1 and 10. Greater or equal to 1, and less than 10.
The second number is called the base. It must always be 10 in scientific notation. In the equation above, the number -8 is the exponent, or in other words, the power of 10.
So how do we convert units into scientific notation?
For example:
NOW WHAT?
It's actually pretty simple.
Make sure you cancel out units as you go!
So now you're left with 9.2 km3 and 103.
But wait! 103 is the denominator! You can't divide 9.2km3 into 103.
So to bring 103 up to the top, you have to make the exponent negative.
Your final answer should be:
See? It wasn't too bad. As you practice, you'll just keep getting better and better.
"Take your time, don't rush, and you'll be fine." -Ms. Chen
Still not getting it? Try watching this useful video on scientific notation.
Subscribe to:
Comments (Atom)






















